  
The difference between the products and the reactants equals negative 268 J/K for the standard entropy change of the combustion of ethylene. The net entropy of the products equals 567.6 J/K, and the net entropy of the reactants is 835.4 J/K. Substituting the values for molar entropies of reactants and products from the reference table yields −. Note that, unlike standard enthalpies of formation of elements, which are zero, standard molar entropies of all substances are greater than zero at 298 K. The standard entropy change for the reaction equals the sum of 2 times the standard entropy of carbon dioxide gas and 2 times the standard entropy of water, minus the sum of the standard entropy of ethylene gas and 3 times the standard entropy of oxygen. 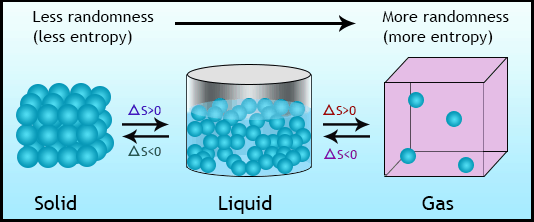
So, like enthalpy changes, entropy changes can be from calculated reference tables of standard molar entropies.įor a reaction occurring under standard conditions, the associated entropy change is determined by the difference between the sum of the standard molar entropies of the products multiplied by their stoichiometric coefficients and the sum of the standard molar entropies of the reactants multiplied by their stoichiometric coefficients.Ĭonsider the combustion of ethylene under standard conditions, where 1 mole of ethylene gas reacts with 3 moles of oxygen gas to produce 2 moles of carbon dioxide gas and 2 moles of water. Enthalpy changes associated with a chemical reaction can be measured with a calorimeter, but the entropy change associated with a reaction cannot be directly measured.Įntropy is a state function, which means that the change in entropy depends solely on the initial and final states of a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
